|
Let us know use these basic principles to account for and compare spectra produced by different types of astronomical objects. The spectrum formed is an emission or bright line spectrum, as shown by the middle spectrum in Figure 1. As these photons can re emitted in any direction an external observer will detect light at these wavelengths. When they de-excite they emit photons of specific frequency and wavelength. If this cloud can be excited by a nearby source of energy such as hot, young stars or an active galactic nucleus then the electrons in atoms of the gas cloud can get excited. 
Stellar spectra typically look like this.Įmission spectrum: A third possibility occurs if an observer is not looking directly at a hot black body source but instead at a diffuse cloud of gas that is not a black body. This means that the resultant spectrum will show dark absorption lines or a decrease in intensity as shown in the dips in the absorption spectrum top right in the diagram above. 
The net effect of this is that the intensity of light at the wavelength of that photon will be less in the direction of an observer. The direction of this re-emission however is random so the chances of it travelling in the same path as the original incident photon is very small. Eventually the electron will de-excite and jump down to a lower energy level, emitting a new photon of specific frequency. Photons of specific frequency can be absorbed by electrons in the diffuse outer layer of gas, causing the electron to change energy levels. The photons emitted from the core cover all frequencies (and energies). If we were able to view the light from this source directly without any intervening matter then the resultant spectrum would appear to be a continuum as shown bottom left in the Figure 1 above.Ībsorption spectrum: Most stars are surrounded by outer layers of gas that are less dense than the core. 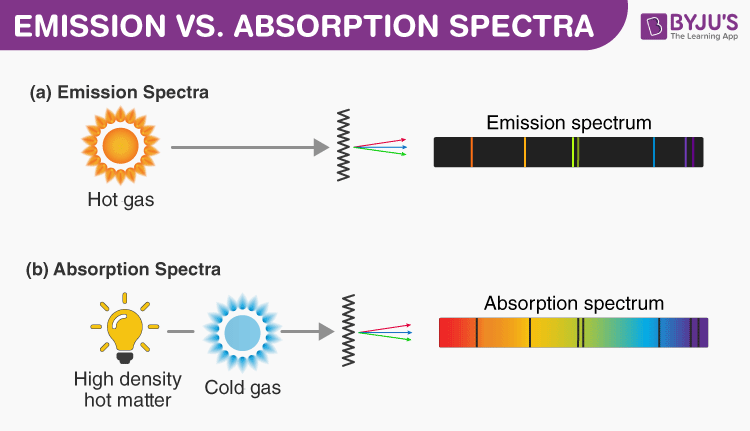
It is the basic unit of all light carrying the energy E= hf.Ītomic emission spectrum: is the pattern of lines corresponds to a different electron transition from a higher energy state to a lower energy state.Figure 1: How continuous, emission and absorption spectra can be produced from same source.Ĭontinuum spectrum: In this diagram, a dense hot object such as the core of a star acts like a black body radiator. Photon: the smallest discrete amount of electromagnetic radiation. Every element has a unique atomic absorption and emission line spectrum.Įxcited-state: of an atom is a state where its potential energy is higher than the ground state.The atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains.In the second example below, where an element has 3 excited states, it could emit photons at 6 specific wavelengths/frequencies (ΔE= hf). Every element has a unique atomic absorption and emission line spectrum. An atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains.Įach of these spectral lines corresponds to a different electron transition from a higher energy state to a lower energy state. When a narrow beam of this light was viewed through a prism, the light was separated into four lines of particular wavelengths. Scientists studied the distinctive pink color of the gas discharge created by hydrogen gas. Signs of other colors contain different gases or mixtures of gases. However, only signs that glow with the red-orange color seen in the figure are filled with neon. 
“Neon” signs are familiar examples of gas discharge tubes. Electrons in the gaseous atoms first become excited, and then fall back to lower energy levels, emitting light of a distinctive color in the process. These gas discharge tubes are enclosed glass tubes filled with a gas at low pressure through which an electric current is passed. Since the electron energy levels are unique for each element, every gas discharge tube will glow with a distinctive color depending on the identity of the gas. One way for atoms to gain energy is to pass an electric current through an enclosed sample of a gas at low pressure called a gas discharge tube.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
